Trump signs executive order accelerating research into psychedelic drug therapies
President Donald Trump signed an executive order Saturday that aims to accelerate research and approval of psychedelic-based therapies, especially drugs like ibogaine, for conditions such as PTSD, depression and addiction.
Trump was joined by Health and Human Services Secretary Robert F. Kennedy, Jr., Centers for Medicare & Medicaid Services Administrator Dr. Mehmet Oz, Food and Drug Administration Commissioner Marty Makary and the podcaster Joe Rogan in the Oval Office for the announcement.
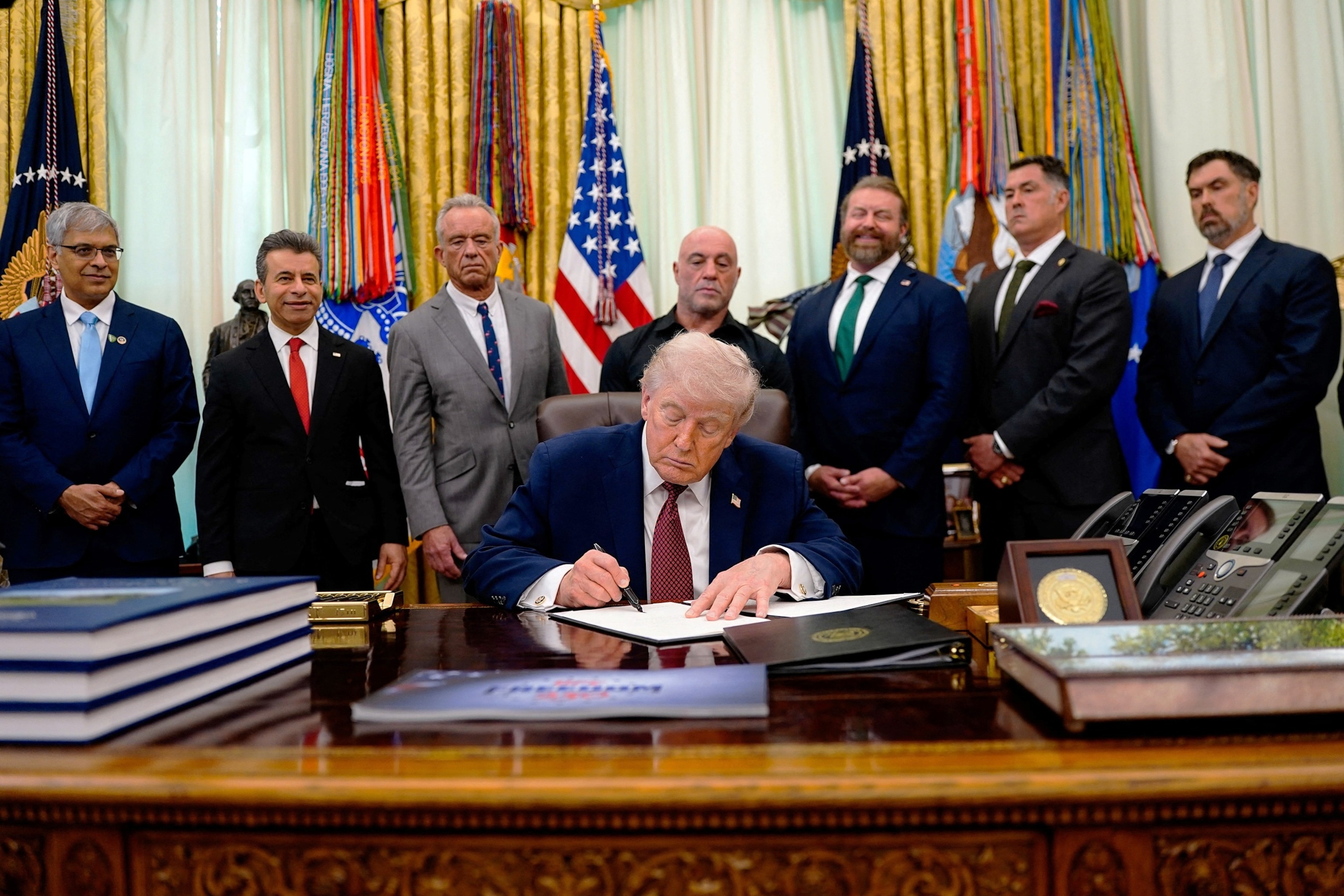
President Donald Trump signs an executive order about easing restrictions on mental health treatments, including, ibogaine, in the Oval Office of the White House in Washington, D.C., April 18, 2026.
Nathan Howard/Reuters
The president said the executive order’s goal was to help veterans with severe mental health issues and provide them with new options for treatment.
“In many cases, these experimental treatments have shown life changing potential for those suffering from severe mental illness and depression, including our cherished veterans, our veterans are having a tremendous hard time,” Trump said.
The president indicated that Rogan lobbied him privately on the issue.
“I got a call from a number of people, including the great Joe Rogan, and he said, we have to do something about this. And I looked into it,” Trump said.
“I went to Bobby and Oz and went to some of the people that work for you, real pros. And everybody came back with the same answer,” the president added.

President Donald Trump signs an executive order in the Oval Office of the White House, on April 18, 2026, in Washington, D.C.
Julia Demaree Nikhinson/AP
Ibogaine is currently classified as a schedule 1 controlled substance, which labels it as not currently accepted for medical use. It has a high potential for abuse, according to the U.S. Drug Enforcement Agency.
Ibogaine, a plant-based psychoactive compound, has limited research for depression, PTSD, anxiety, and traumatic brain injury, with most evidence coming from animal studies and small, early human studies with major limitations. While animal studies are an important part of research they aren’t the same as human trials and results often don’t translate for humans.
There is some evidence suggesting that ibogaine carries serious safety risks, particularly a dangerous irregular heart rhythm known as arrhythmias which can be fatal, along with neurologic and gastrointestinal side effects.
Psilocybin, the active compound in so-called magic mushrooms, has the strongest evidence so far for depression and has received Breakthrough Therapy designation from FDA for treatment-resistant cases.
It has been de-criminalized in a handful of states but remains a class 1 controlled substance federally.

President Donald Trump sits, on the day he signs an executive order about easing restrictions on mental health treatments, including ibogaine, as National Institutes of Health (NIH) Director Jay Bhattacharya, Food and Drugs Administration (FDA) head Marty Makary, U.S. Health and Human Services Secretary Robert F. Kennedy Jr., podcaster Joe Rogan, Americans for Ibogaine CEO W. Bryan Hubbard, Rep. Morgan Luttrell and his brother Marcus Luttrell, a retired U.S. Navy SEAL, stand, in the Oval Office of the White House in Washington, D.C., April 18, 2026.
Nathan Howard/Reuters
In a recent Nature Medicine review of 12 studies that evaluated the effect of psilocybin, when combined with psychotherapy or psychological support, each study showed that psilocybin significantly reduced depression symptoms with rapid and consistent effect over several weeks when compared to control conditions.
Beyond symptom reduction, patients receiving psilocybin were significantly more likely to achieve meaningful clinical improvement — response rates were nearly three times higher, and remission rates were about four times higher than the control groups.
MDMA has shown promise for PTSD but was not FDA approved in 2024 despite that same Breakthrough designation due to concerns about how the clinical trials were conducted, inconsistent results, safety concerns, and requested additional research before they would re-review, ABC news has previously reported.
It is also a class 1 controlled substance.
These treatments still require careful monitoring in controlled settings by medical professionals. Researchers have limited data on their long-term side effects or success.
Even with some signs of early promise using there are major safety concerns and uncertainty about how well the benefits would stand up in real world settings. Current studies and research focus on the most severe forms of these mental health conditions that are resistant to existing treatment.

President Donald Trump holds up a signed executive order in the Oval Office of the White House, on April 18, 2026, in Washington, D.C.
Julia Demaree Nikhinson/AP
The president noted that the U.S. has been researching the substances and claimed that the order would clear out “unnecessary bureaucratic hurdles.”
Trump also said that the order provides for expedited rescheduling for any psychedelics that later receive approval from the FDA for treatment.
“It directs the FDA to expedite their review of certain psychedelics already designated as breakthrough therapy drugs. They’re very much, being discussed. It’s one of the hottest things. I think you’re talking about Oz, right?” Trump said.
“If these turn out to be as good as people are saying, they’re going to have a tremendous impact on this country and other countries, too,” he added.
Kennedy has been critical of the use of antidepressants and other current therapies used to treat these mental health conditions.